Upcoming changes in 2026 EU/UK

It's another BIG year for cosmetic regulatory changes.
80+ Fragrance allergens
EU only
Key date: 31 July 2026
We've had 5 years to prepare for this but it somehow still feels like it is too soon. The existing 26 allergens which need to be declared on the label in cosmetic products.
All new products placed on the marketplace after 31 July 2026 MUST have the new declarable allergens on the labelling.
When fragrance allergen concentration exceeds:
— 0,001 % in leave-on products
—0,01 % in rinse-off products.
Full list of new fragrance allergens can be found here:
https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32023R1545
Actions:
- Reach out to your fragrance and essential oil suppliers for updated allergen lists.
- Some extracts and active ingredients are now disclosing trace amounts of fragrance allergens - check that you are not affected.
- Update your labels! New products released after 31 July 2026 MUST have declarable allergens placed on the label. After 31 July 2028 all products in the EU must declare their extended the new declarable allergens on the labelling.
- Update your Product Information File (PIF), you may also need to update your Cosmetic Product Safety Report (CPSR).
Warning! I have noticed that some IFRA declarations (International Fragrance Association) are now declaring other fragrance ingredients which were not disclosed previously. This is a problem as these ingredients have been restricted since 2013, but you may not be aware it was present in your product. Check that your products are still legally compliant!
An example of this is Methyl octine carbonate which is allowed at a maximum of 0,002 % when used alone. When present in combination with methyl heptine carbonate, the combined level in the finished product shall not exceed 0,01 % (of which methyl octine carbonate shall not be more than 0,002 %). I'm seeing a few IFRA certificates which do not comply at the moment so be careful.
Remember - Swift Fox Consultancy Ltd provides horizon scanning, ingredients listings as part of the CPSR service. Let us take the headache out of compliance.
Tea Tree Oil (TTO)

Region: EU
Key date: Not yet confirmed
The first in our list of regulatory 'disagreements' between the EU and UK. But...while the implementation date has not been set, tea tree oil is such a widely used ingredient, I think it is wise to prepare for the changes ahead of time.
Luckily for the UK, there seems to be no need to change - yet.
| UK | EU | |
| REACH status | Disagrees with EU opinion - not classed as a developmental toxin | Classed as CMR 1B (reprotox) |
| Cosmetic regulatory changes | No change | Tea tree oil must be International Standard (ISO 4730:2017) standard. Recommended use levels. 2% in shampoo 1% in shower gel 1% in face wash 0.1% in face creams |
Actions:
- Check that your supplier(s) comply with International Standard (ISO 4730:2017).
- Prepare to reformulate if you exceed the SCCS recommended levels.
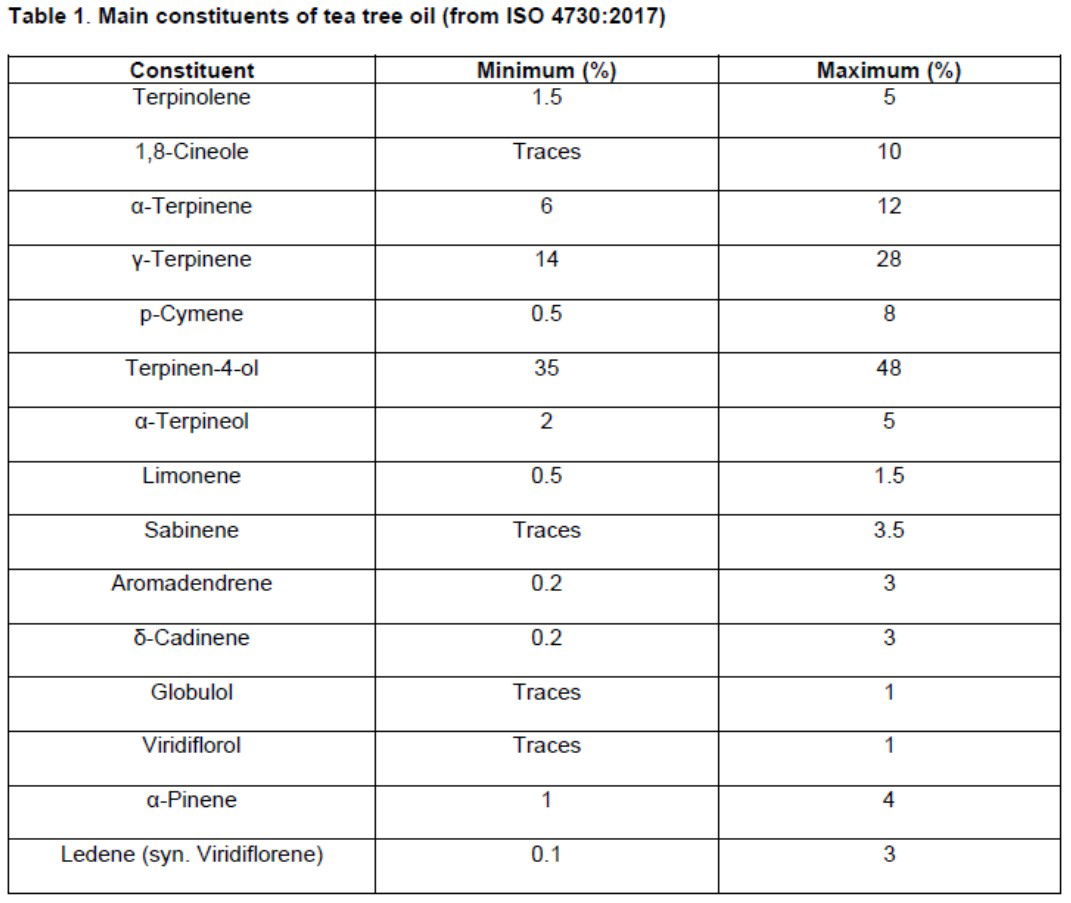
Examples of some of the requirements of ISO 4730:2017.
Citral
Region: EU
Key dates: From 1 January 2027 cosmetic products containing these substances and not complying with the restrictions shall not be placed on the Union market. From 1 July 2028 cosmetic products containing these substances and not complying with the restrictions shall not be made available on the Union market.
This is a big one. Citral is in so many essential oils and fragrance oils, giving a fresh citrus scent.
Essential oils which are high in citral:
* Lemongrass
* May chang
* Lemon balm
* Citrus oils

While it is not in the EU regulatory text yet - we have a early access version that was submitted to the WTO:
So we have a very good idea of the dates and restrictions that the EU are planning on implementing.
The UK has not assessed citral yet, but is expected to assess citral in 2026.
| Product | Maximum use | |
| Citral / Geranial / Neral | Make-up products, lipstick, lip salves | 0.11 % |
| Solid deodorants and anti-perspirants | 0.032 % | |
| Eye products, face make-up and make-up remover | 0.65 % | |
| Fragrance products (hydroalcoholic and non-hydroalcoholic | 0.6 % | |
| Leave-on skin products (except lip products, solid deodorants and antiperspirants, fragrance products, eye products, face make-up and makeup remover | 0.15 % | |
| Oral products | 0.35 % | |
| Hair leave-on products | 1.2 % | |
| Skin and hair rinse-off products | 1.2 % | |
| Leave-on products for the anogenital area (baby wipes and intimate wipes | 0.063 % | |
| Essential oils which are high in citral: Lemongrass, May chang, citronella, citrus oils | ||
Actions:
- Check your products which contain fragrance oils or essential oils which contain citral.
- Reformulate if necessary.
Benzyl Salicylate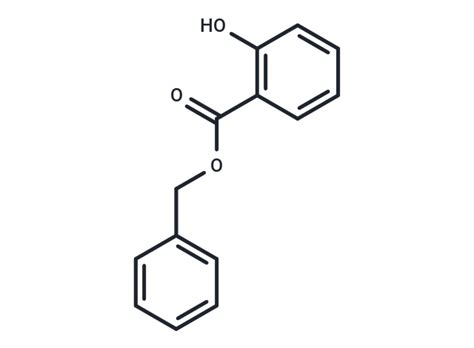
Region: EU
Key dates: From 1 January 2027 cosmetic products containing these substances and not complying with the restrictions shall not be placed on the Union market. From 1 July 2028 cosmetic products containing these substances and not complying with the restrictions shall not be made available on the Union market.
While it is not in the EU regulatory text yet - we have a early access version that was submitted to the WTO:
So we have a very good idea of the dates and restrictions that the EU are planning on implementing.
| Product | Maximum use | |
| Benzyl salicylate | Hydro-alcoholic based fragrances (spray and non-spray) | 4 % |
| Rinse-off skin and hair products (except rinse-off body products) | 0.5% | |
| Rinse-off body products | 1.3% | |
| Leave-on skin and hair products (non-spray/non-aerosol) (except body lotion) | 0.5% | |
| Leave-on hair products (spray/aerosol) | 0.5% | |
| Leave-on body products (nonspray/spray/aerosol | 0.7% | |
| Face make-up and make-up remover products | 0.2% | |
| Oral products | 0.004% | |
| Deodorant products (spray/aerosol) | 0.91% |
Methyl salicylate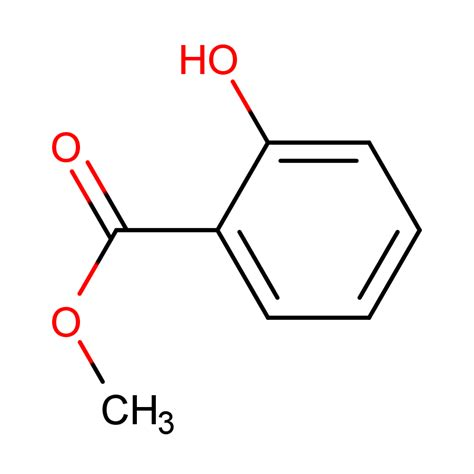
Region: EU + UK
Key dates:
EU - 31 July 2026
UK - 31st March 2026
We'll have quite a few examples in todays blog of EU and the UK having a difference of opinion. Methyl salicylate is a fragrance ingredient that is also found naturally in oil of wintergreen. It is also used as a flavouring agent in foods but at very low levels.
The SCCS reviewed methyl salicylate after its classification as Classed as CMR 2B (reprotox). In particular they were concerned about potential effects on babies and young children.
The UK have taken a stricter stance than the EU.
This is a particular risk if you are a brand owner of cosmetic products designed to be used on babies and young children.
| Methyl Salicylate: UK/EU cosmetic regulations | ||
| EU | UK | |
| Implementation date | 31 July 2026 | 31st March 2026 |
| Leave-on skin products (except face makeup, spray/aerosol body lotion, spray/aerosol deodorant and hydro alcoholic-based fragrances) and leave on hair products (except spray/aerosol products) | 0.06% | 0.02% (products intended for children 0.5-1 years) 0.06% (products intended for children above 1 year and adults) |
| Face makeup (except lip products, eye makeup and makeup remover) | 0.05% | 0.05% |
| Eye makeup and makeup remover | 0.002% | 0.002% |
| Leave-on hair products (spray/aerosol) | 0.009% | 0.009% |
| Deodorant spray/aerosol | 0.003% | 0.003% |
| Body lotion spray/aerosol | 0.04% | 0.04% |
| Rinse-off skin products (except hand wash) and rinse-off hair products | 0.06% | 0.02% (products intended for children 0.5-1 years) 0.06% (products intended for children above 1 year and adults) |
| Hand wash | 0.6% | 0.02% (products intended for children 0.5-1 years) 0.6% (products intended for children above 1 year and adults) |
| Hydroalcoholic-based fragrances | 0.6% | 0.6% |
| Lip products | 0.03% | 0.02% (products intended for children 0.5-1 years) 0.03% (products intended for children above 1 year and adults) |
| Toothpaste | 2.52% | 2.5% |
| Mouthwash intended for children aged 6–10 years | 0.1% | 0.1% |
| Mouthwash intended for children above 10 years of age and adults | 0.6% | 0.4% |
| Mouth spray | 0.65% | 0.65% |
Highlighted cells show where regulatory limits differ.
Actions:
- Review your formulations for Methyl Salicylate.
- Consider using age warnings on your product labelling.
- Reformulate according to where you are selling your product and the age of the youngest consumer likely to use your product.
Vetiveria Zizanioides Extract
Region: EU
Key dates: From 1 January 2027 cosmetic products containing these substances and not complying with the restrictions shall not be placed on the Union market. From 1 July 2028 cosmetic products containing these substances and not complying with the restrictions shall not be made available on the Union market.
While it is not in the EU regulatory text yet - we have a early access version that was submitted to the WTO:amending Regulation (EC) No 1223/2009 of the European Parliament and of the Council as regards the use of Benzyl Salicylate, Triphenyl Phosphate, Ammonium Silver Zinc Aluminium Silicate, Aluminium, water-soluble zinc salts, Acetylated Vetiver Oil, Citral, HC Blue No. 18, HC Red No. 18, HC Yellow No. 16, Hydroxypropyl pphenylenediamine and its dihydrochloride salt, and DHHB in cosmetic products
So we have a very good idea of the dates and restrictions that the EU are planning on implementing.
| Product | Maximum use | |
| Vetiveria Zizanioides Root Extract; Vetiveria Zizanioides Root Oil; Vetiver | Fragrance | 0.9% |
| Deodorant | 0.05% | |
| Make-up products | 0.05% | |
| Leave-on products | 0.1% | |
| Rinse-off products | 0.2% |
Actions:
- Review products containing vetiver to check if they comply.
- Reformulate if necessary.



